Easily Secure CDSCO Medical Device Loan License in India
Hassle‑free CDSCO medical device loan license application services
End‑to‑end support for easy compliance with CDSCO requirements, thereby helping you avoid delays and achieve faster product launch in the Indian market.
Complete CDSCO loan license application support
Expertise in medical devices & IVDs
Faster approvals with compliant submission
Trusted partner for manufacturers seeking CDSCO loan license
600+ Granted Approvals 27+ Years Experience
300+ Happy Clients












The CDSCO grants the medical device loan license to help medical device manufacturers not having a manufacturing facility or infrastructure. Notably, for obtaining a CDSCO loan license, you have to navigate through the regulatory procedures, including documentation and quality assurance protocols. Importantly, compliance with legal and safety standards is mandatory. This is necessary to ensure although the manufacturing is outsourced, the responsibility for the quality remains with the licensee. Thus, the medical device loan license applicant must meet all the regulatory criteria set by CDSCO. Pharmadocx Consultants is a renowned medical device regulatory consultant. We leverage our expertise and industry knowledge to help medical device manufacturers easily secure the CDSCO loan license. Our expertise will help you avoid the common pitfalls encountered while securing the CDSCO loan license for medical devices.
Securing CDSCO loan license for medical devices
The CDSCO loan license has certain regulatory requirements that have to be fulfilled. Securing the CDSCO medical device loan license is not an easy task. You need expertise in the regulatory guidelines and need to have a clear understanding of the regulations. Furthermore, the license application process is tedious and complicated.
With the help of our team of regulatory experts, you can easily secure the CDSCO loan license for medical devices. Our comprehensive service includes document preparation, license application, and continuous support till you successfully secure the license. The Pharmadocx Consultants team will help you navigate through the complex regulatory landscape to streamline the license acquisition process.
Our commitment to excellence and in-depth understanding of regulatory requirements makes us a trusted partner for manufacturers seeking CDSCO loan licenses. Let us help you have a smooth sailing CDSCO regulatory journey.
What is the CDSCO medical device loan license?
The CDSCO medical device loan license is granted to a medical device manufacturer, not having a production facility, who wants to utilize the manufacturing facility/site of another manufacturer. It is granted only if the “other manufacturer” also manufactures the same medical device at the facility as the loan licensee. There should be a mutual agreement between two parties for utilizing the manufacturing infrastructure to produce the same medical device. In order to apply for a loan license, the licensee must select a manufacturer with a valid CDSCO manufacturing license.
Criteria for applying for CDSCO medical device loan license
- The medical device manufacturing team should consist of qualified technical personnel with expertise in medical device testing. The personnel are required to oversee and supervise the medical device manufacturing and testing process.
- The manufacturing facility must comply with all applicable quality management system guidelines.
License forms
- Class A and B medical devices: MD-6
- Class C and D medical devices: MD-10
Benefits of the medical device loan license scheme
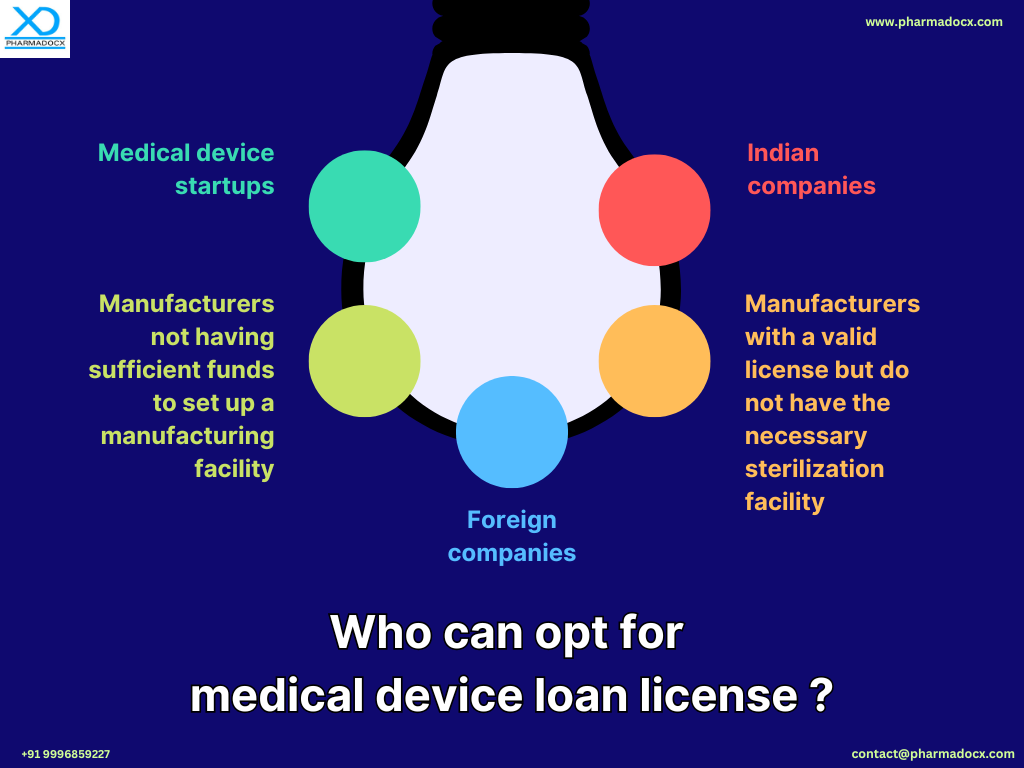
The medical device loan license can help support and boost the medical device manufacturing sector. This scheme will support manufacturers by helping them save funds that would have been spent on setting up manufacturing facilities.
- Manufacturers not having manufacturing facilities/infrastructure can benefit from the loan license scheme.
- Small firms not having sufficient funds to invest on manufacturing units can enter the medical device manufacturing industry.
- The manufacturer can allocate the funds intended for manufacturing facilities or equipment to research and innovation.
- Manufacturers facing constraints due to insufficient production capacity can considerably benefit from the loan license scheme.
- Instead of constructing, managing, and maintaining, a manufacturing facility, the medical device manufacturer can focus on market expansion.
Licensing authorities for different CDSCO medical device classes
CDSCO has categorised medical devices into various classes depending on their intended use and risk level. This medical device classification system helps with the CDSCO registration process. Moreover, the license application process and licensing authorities vary depending on the CDSCO class to which the medical device belongs to. Hence, it is important to know which form to use while applying for the loan license. We have provided an overview of the license application process, licensing authorities, and necessary forms:
Class A and B medical devices
To secure a CDSCO medical device loan license for Class A and B medical devices, you need to apply to the State Licensing Authority (SLA). Apply online using the Form MD-4 and pay the necessary processing fee. The regulatory body will relay the approval via Form MD-6.
Class C and D medical devices
To secure a CDSCO loan license for Class C and D medical devices, you need to apply to the Central Licensing Authority (CLA). Apply online using the Form MD-8 and pay the necessary processing fee. Then, the regulatory officers will inspect the production facility within 60 days from the date of application. After which, they will relay the approval via Form MD-10.
To check which CDSCO class your medical device belongs to, use our free tool.
Necessary documents for CDSCO medical device loan license
We have provided an overview of the documents required for applying for CDSCO loan license for medical devices:
- Cover letter
- TR6 challan
- All applicable forms
- Declaration of qualified technical personnel who will be involved in the medical device manufacturing and testing process
- Plant master file
- Device master file
- Quality Management System documents
- Medical device manufacturing license
Do you want to secure your CDSCO medical device loan license in a hassle-free manner?
Fill out the form below
Key CDSCO medical device loan license details
- The loan licensee will be responsible for compliance with necessary quality and safety standards for the medical device being manufactured.
- The licensor will provide the licensee with the necessary manufacturing facility and required infrastructure for manufacturing the device. The licensor will share the technical know-how for manufacturing the medical device.
- The CDSCO loan licensee must pay a fee for using the licensor’s medical device manufacturing facility and infrastructure.
- The licensee must secure the required CDSCO regulatory approvals to manufacture and market their medical device in India.
Validity of CDSCO loan license for medical devices
The medical device loan license remains valid for an indefinite period. However, a license retention fee needs to be paid every 5 years. The fee amount will depend on the medical device class. Notably, the license will remain valid indefinitely only if it is not cancelled or suspended.
Why choose Pharmadocx Consultants?
Medical Device Licences
Years Experience
Plants Set-up
Our Clients

How can Pharmadocx Consultants help you obtain the CDSCO medical device loan license?
The loan license for medical devices allows manufacturers not owning a manufacturing facility to manufacture medical devices. The CDSCO loan license has certain regulatory requirements that the applicant has to fulfil. Furthermore, various supporting documents have to be prepared. Hence, the loan license application process is cumbersome. We at Pharmadocx Consultants offer comprehensive CDSCO loan license application service.
- Document preparation: We will help you prepare the necessary documents for license application.
- Loan license application: Our team will provide guidance and help you fill in the correct application form. We will walk you through the complex license application process.
- Mock audit: We conduct mock audits to better prepare you for the regulatory inspection and audit. Moreover, we not only identify lapses but also provide actionable next steps.
- Ongoing license grant support: Our support does not end with the license application. We provide continuous support till you successfully secure the license.
We specialize in streamlining the complex process of CDSCO medical device loan license application. Our team of experts will be more than happy to help you secure the loan license in a hassle-free manner.
Let's Talk!
We'd love to hear from you! Whether you have questions about our pharmaceutical plant setup consultation services or want to discuss a potential project, our team is here to help. Simply fill out the form below, and we'll get back to you as soon as possible. Alternatively, you can reach out to us directly using the phone number or email address listed on this page. We look forward to connecting with you!
Phone / Whatsapp
Address
- Head Office - Opposite Dewan Mill, Old D.C. Road Sonepat - 131001 Haryana, India
- Registered Office - Netaji Subhash Place, Delhi, 110034

