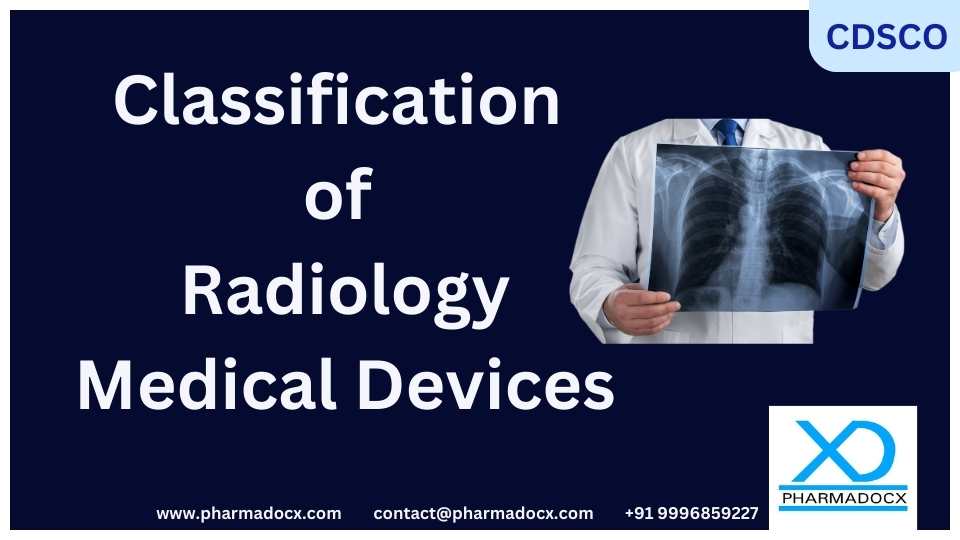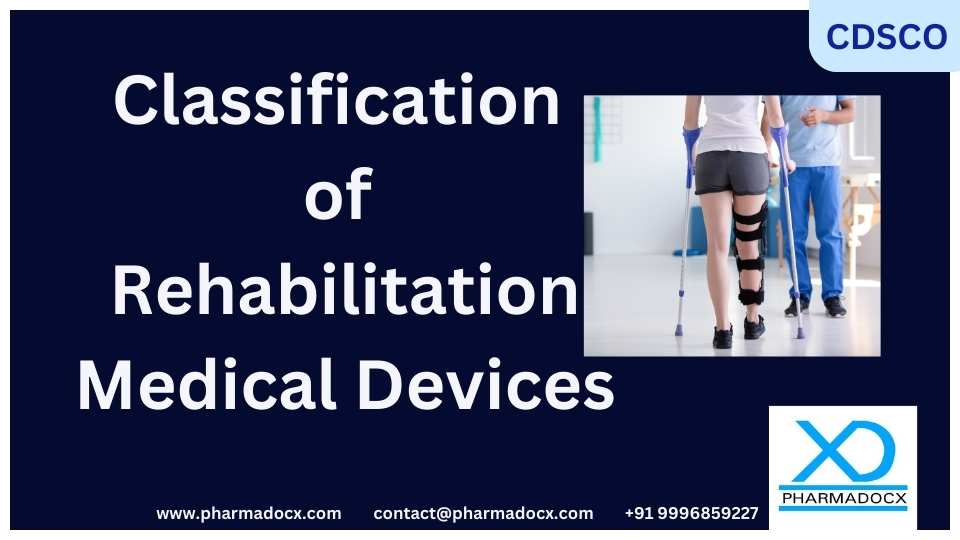
CDSCO Classification of Rehabilitation Medical Devices
The Central Drugs Standard Control Organisation (CDSCO) is the regulatory authority for the medical device industry in India. On 13 September 2021, CDSCO specified 60 Rehabilitation medical devices. This list includes devices such as Exothermic heat therapy packs,...
CDSCO Classification of ENT Medical Devices
The Central Drugs Standard Control Organisation (CDSCO) is the regulatory authority for the medical device industry in India. On 13 September 2021, CDSCO specified 67 ENT medical devices. This list includes devices such as Nasal septum straightening forceps, ENT...
CDSCO Sugam Portal: A Comprehensive Guide
The Central Drugs Standard Control Organization (CDSCO) is India’s primary regulatory body for ensuring the safety, efficacy, and quality of drugs, cosmetics, and medical devices. To streamline its operations and make the process of applications and approvals...