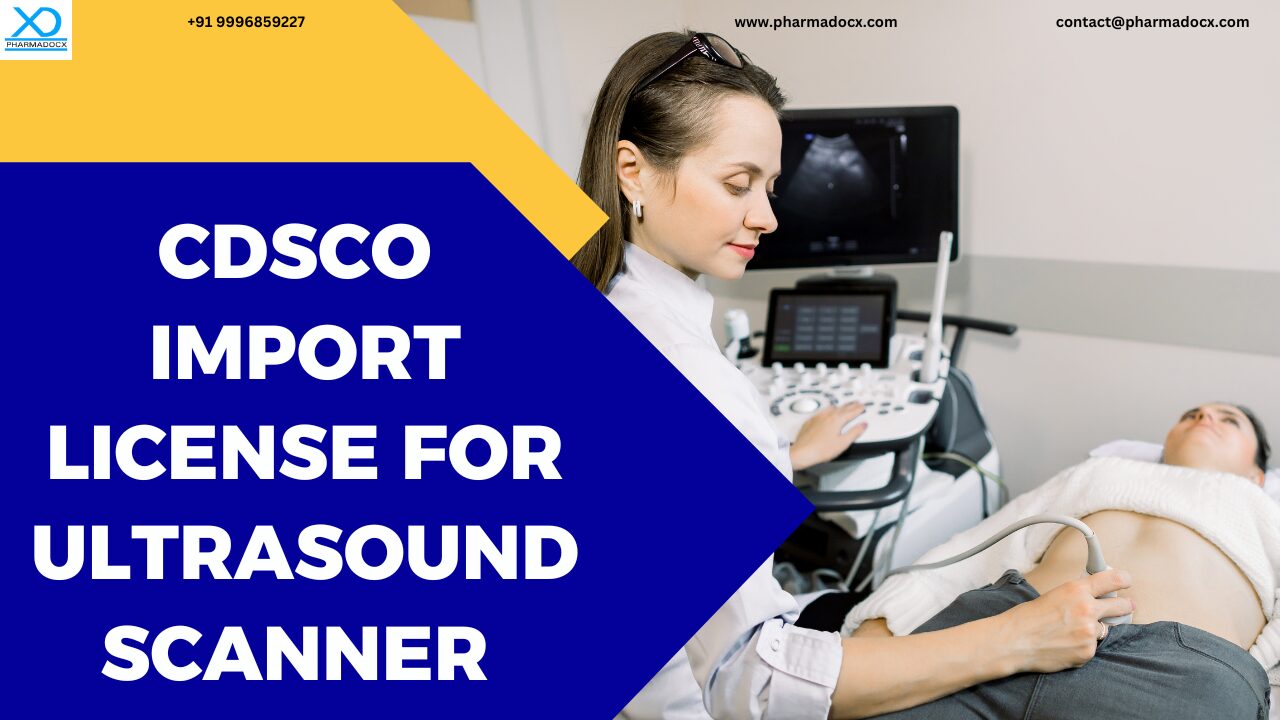
India still remains heavily dependent on imports for ultrasound scanners. Most high-end systems are sourced from multinational companies. Domestic manufacturing is limited to basic and mid-range devices, leaving advanced imaging technologies to be mostly import-driven. CDSCO, the apex medical device regulatory body, oversees and regulates the import of ultrasound scanners. Hence, to import ultrasound scanners, you need to mandatorily comply with the CDSCO medical device regulations. Additionally, you have to secure the CDSCO import license for ultrasound scanner to launch your product in the Indian market.
India’s ultrasound scanner market
India’s ultrasound scanner market is import-heavy for most advanced systems. Domestic manufacturers remain focused on low-cost portable and entry-level systems. Thus, most ultrasound scanners in India are imported. The growth of India’s imported ultrasound market is driven by rising demand for advanced imaging technologies, expanding applications in cardiology and oncology, and the lack of domestic R&D capacity for high-end devices. Imports now account for over 70% of the market, with multinational firms dominating hospital and diagnostic center procurement.
Key factors driving the growth of India’s imported ultrasound market
We have highlighted some of the key factors driving the growth of India’s imported ultrasound market:
- Expanding clinical applications: Obstetrics and gynecology are still the largest segment, with demand for 3D/4D fetal imaging. In cardiology and oncology, there is a growing use of Doppler and high-resolution ultrasound for vascular and tumor imaging. Moreover, portable imported devices are being increasingly used in ICUs and emergency medicine.
- Technological advancements: Imported systems offer superior resolution (3D/4D imaging) and real-time visualization. Notably, automated measurements, anomaly detection, and workflow optimization are available only in imported devices. Moreover, high-intensity focused ultrasound (HIFU) used for non-invasive tumor therapy are largely unavailable in domestic systems.
- Hospital and diagnostic center demand: Tier-1 and Tier-2 city hospitals prefer globally validated systems for compliance and patient trust. Private diagnostic chains heavily rely on imported scanners to maintain competitive service quality.
- Regulatory and quality considerations: Imported devices carry international approvals, making them more attractive for hospitals under audit pressure.
- Economic and market factors: India’s healthcare market is expanding, thereby boosting demand for advanced imaging. Hospitals catering to international patients prefer imported systems to meet global expectations. Furthermore, while imports are costlier, hospitals still prioritize advanced features over price.
Thus, there is a high demand for imported ultrasound scanners in India. To import ultrasound scanners in India, you have to mandatory secure the CDSCO medical device import license. We have prepared this guide to help you easily secure the CDSCO import license for ultrasound scanner.
CDSCO import license for ultrasound scanner
- Form MD 14: To import ultra sound scanners into India, an application has to be filed under Form MD 14.
- MD 15 license: The license for importing ultrasound scanners for sale and distribution in India is granted under the CDSCO MD 15.
Documents for CDSCO import license for ultrasound scanner
Supporting documents have to be prepared for the CDSCO import license application. These documents have a vital role in demonstrating regulatory compliance.
- Self-attested copy of valid manufacturing license demonstrating the device is manufactured in accordance with the regulatory requirements of the country of origin.
- Plant master file
- Device master file
- Notarized copy of overseas manufacturing site or plant registration in the country of origin issued by the competent authority
- Notarized copy of ISO 13485 certificate of the actual medical device manufacturer
- Duly apostilled/notarized copy of Free Sale Certificate from National Regulatory Authority of country of origin
- Full Quality Assurance Certificate/CE type examination certificate/CE product quality assurance certificate
- Notarized Declaration of Conformity stating the device complies with applicable regulatory requirements and standards.
- A copy of the latest inspection or audit report carried out by Notified bodies or the National Regulatory Authority within last 3 years
- Power of attorney, in case an authorized agent has to be appointed
- Constitution details of Indian authorized agent for foreign medical device manufacturers.
- Notarized CE design certificate
For a detailed list of necessary supporting documents, feel free to get in touch with us. Furthermore, we provide document preparation support. Our team will not only prepare the documents but also compile them per CDSCO regulatory guidelines. Accurate and correct documentation per guidelines is critical to easily get the CDSCO import license for ultrasound scanner.
Rules for foreign medical devices manufacturers
Foreign medical device manufacturers, not having a local presence in India, planning to sell their devices in India will have to appoint an authorized agent. The authorized agent must have a valid wholesale license (MD 42). Thus, the Indian authorized agent will file the CDSCO medical device import license application on behalf of the foreign manufacturer. Furthermore, the authorized agent will act as a liaison between the foreign manufacturer and CDSCO.
Who can apply for the CDSCO MD 15 license?
- Any wholesaler or importer who wants to import ultrasound scanner for sale and distribution in India
- Any foreign medical device manufacturer who wants to supply medical devices in India. However, the foreign manufacturer will have to file the application via the authorised agent.
- An Indian authorized agent who has a valid wholesale license for the sale or distribution of medical devices.
CDSCO import license application fees
The CDSCO has formulated the medical device classification system to ease the license application process. The application fees required will depend on the CDSCO medical device class to which your device belongs to. As ultrasound scanners belong to CDSCO Class C, the application fees necessary for a Class C medical device will be required. A fee of$3,000 per site along with $1,5000 per product will be required.
How to get the license to import ultrasound scanners into India?
- Step 1: Register on the official CDSCO online portal, SUGAM.
- Step 2: Prepare and compile all necessary supporting documents per CDSCO guidelines.
- Step 3: Fill in the MD 14 application form with all the required details. Ensure accuracy and completeness.
- Step 4: Upload the duly filed in application form along with the necessary documents on the online portal. Next, pay the necessary fees for the import license application. Once you have successfully completed all the steps and submitted your application, you will receive an application number.
- Step 5: The CDSCO will review the CDSCO import license application for ultrasound scanner for compliance with regulatory guidelines and verify the documents. They will try to check for any areas of non-conformities. It is important to promptly and appropriately respond to all queries. Response should be provided in a timely manner along with additional documents (if required) to support the response.
- Step 6: The CDSCO may inspect the importing firm’s medical device facility to verify whether it meets the quality and regulatory standards.
- Step 7: CDSCO will evaluate all the information provided and check the response to the queries. If satisfied, they will approve the application. The CDSCO medical device import license will be granted under MD 15. This license will permit you to import ultrasound scanners for sale and distribution in India.
Validity of the CDSCO import license for ultrasound scanner
The CDSCO import license for ultrasound scanner remains valid indefinitely. However, a license retention fee has to be paid every 5 years to retain its validity.
Pro tips for smooth CDSCO import license application
- Power of Attorney must be prepared per Medical Device Rules (MDR), 2017
- Product name and model numbers must be consistent across POA, FSC, product label, and other technical documents
- Notarization of documents must follow MDR, 2017, guidelines
- Device master file (DMF), plant master file (PMF), and other technical documents need to be prepared in the format prescribed in MDR, 2017
To easily secure the CDSCO import license for ultrasound scanner, email at [email protected] or call/Whatsapp on 9996859227.